The piRNA pathway
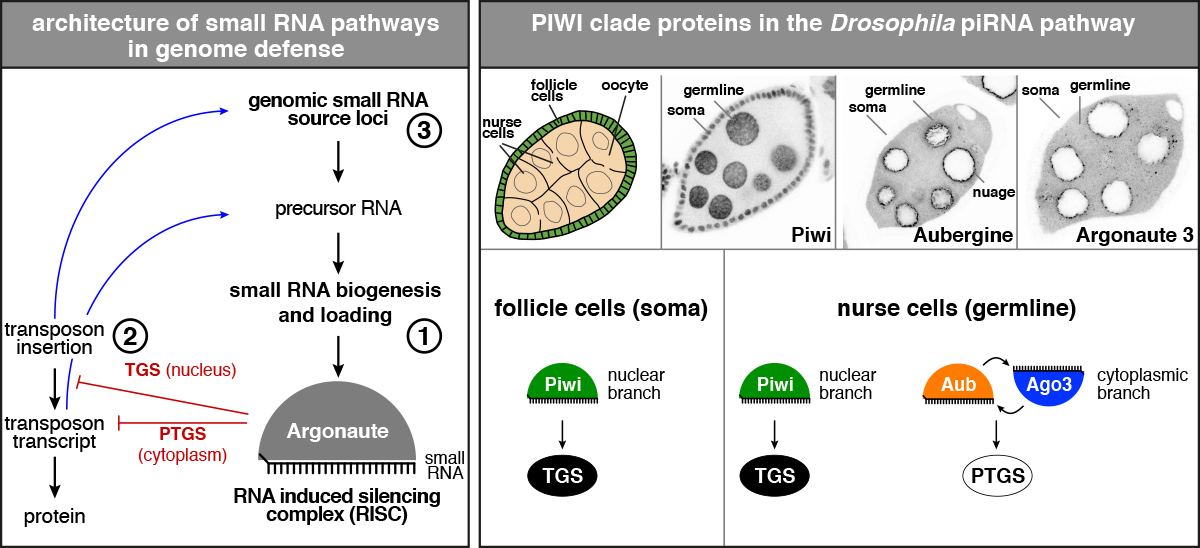
PIWI proteins are the central and name-giving proteins of the piRNA pathway. They represent a distinct subfamily of Argonaute proteins, molecular machines that are loaded with small RNAs, here the PIWI-interacting RNAs (piRNAs). piRNAs act as sequence specific guides to target PIWI proteins to complementary RNAs for their silencing. Based on this, three major conceptual domains within the pathway can be defined:
(1) piRNA biogenesis (programming of the effectors)
(2) piRNA-guided silencing (interference with target gene expression)
(3) biology of the genomic piRNA source loci (memory and adaptation)
Remarkably, the same three core concepts also apply to bacterial CRISPR/Cas systems. RNAi and CRISPR/Cas do not share a common evolutionary origin. We therefore believe that this points to why small RNA based silencing systems have been so successful in genome defense throughout evolution:
By fuelling small RNA biogenesis with transcripts of the targets themselves, these pathways provide flexibility to any nucleic acid sequence and adaptability to new challenges originating from sequence drift or the horizontal invasion of new transposable elements.
Drosophila oogenesis is among the most powerful and influential systems to study the piRNA pathway (reviewed in Senti & Brennecke. 2010). Three PIWI-clade proteins (Piwi, Aubergine, Argonaute 3) are expressed throughout ovarian development, from the embryonic primordial germ cells till the nurse cells of the late egg chambers. Only the nuclear Piwi protein is additionally expressed in somatic support cells of the ovary, where it silences retroviral elements that are capable of hijacking cellular vesicles travelling from follicle cells to the oocyte. From these somatic support cells, a stable cell line has been derived, the OSS/OSC cell line. It represents the only existing cell line that expresses a nuclear PIWI clade protein and is therefore a powerful and unique tool to study piRNA biogenesis and transcriptional target gene silencing.
Using the Vienna transgenic RNAi library, we have conducted genetic screens in the developing ovary to systematically identify piRNA pathway factors in ovarian germ cells and somatic support cells (Handler et al. 2013: The genetic Makeup of the Drosophila piRNA pathway). Based on these screens, ~40-50 proteins are required for the piRNA pathway, some in germline and soma, others specific for either germline or soma. The identified proteins—many of which are entirely uncharacterized—provide us with strong starting points to study the molecular and conceptual architecture of the pathway. The image to the left for example depicts a GFP-tagged piRNA pathway factor that co-localizes with Aubergine and Ago3 in peri-nuclear clouds.
1. piRNA biogenesis – how to distinguish self from nonself
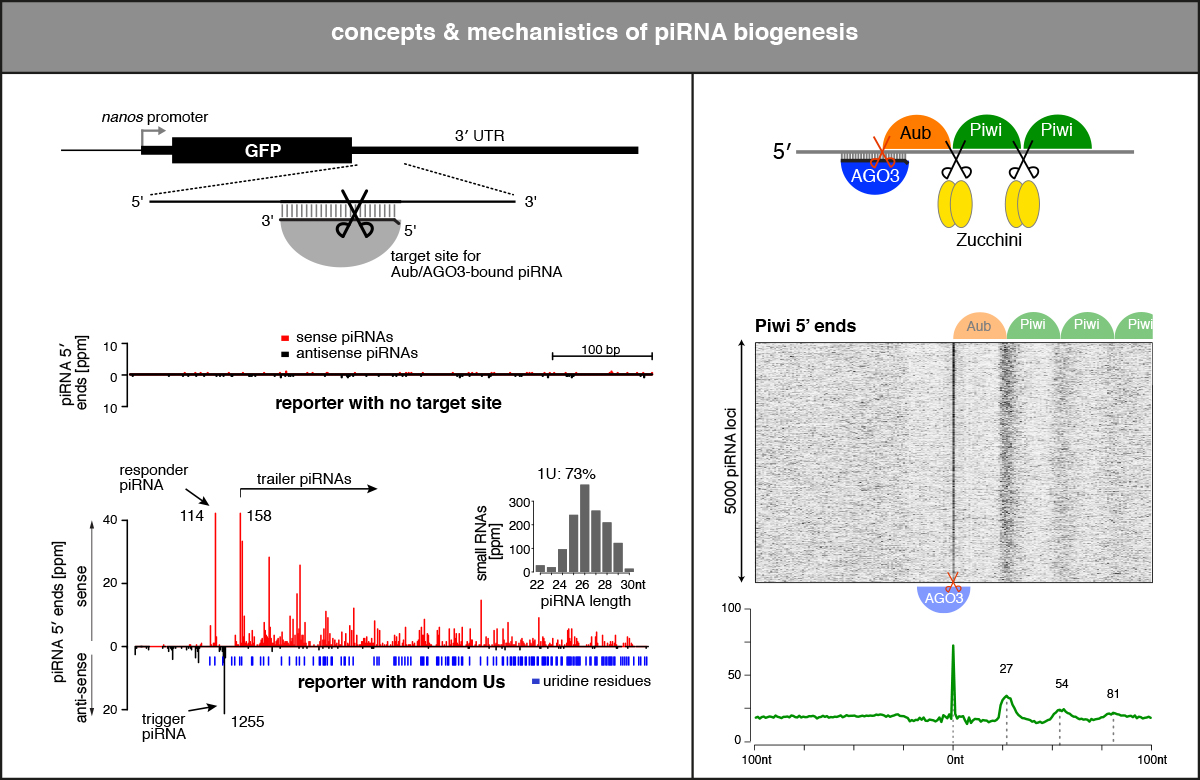
Hundreds of thousands of piRNAs are processed from single-stranded precursor RNAs in a poorly understood manner. More than a dozen proteins—RNA helicases, nucleases, adaptor proteins, chaperones, and several proteins with unknown function—are involved. Curiously, several of these biogenesis factors are inserted into the outer mitochondrial membrane. Using genetics, computational analyses of piRNA populations, and biochemistry, we unravel the rules of piRNA biogenesis and dissect the underlying molecular mechanisms. A major challenge for the future is to reconstitute aspects of piRNA biogenesis in vitro using purified components and single molecule approaches. This is an uncharted and exciting research area for ambitious RNA-protein biochemists. Understanding the molecular details of piRNA biogenesis will be essential to explain how the cell distinguishes non-self from self, and piRNA precursor transcripts from other single-stranded RNAs in the cell.
Relevant literature:
- Discrete small RNA-generating loci as master regulators of transposon activity in Drosophila (Brennecke et al. 2017)
- Specialized piRNA pathways act in germline and somatic tissues of the Drosophila ovary (Malone & Brennecke et al. 2009)
- An in vivo RNAi assay identifies major genetic and cellular requirements for primary piRNA biogenesis in Drosophila (Olivieri et al. 2010)
- A systematic analysis of Drosophila TUDOR domain-containing proteins identifies Vreteno and the Tdrd12 family as essential primary piRNA pathway factors (Handler et al. 2011)
- The cochaperone Shutdown defines a group of biogenesis factors essential for all piRNA populations in Drosophila (Olivieri et al. 2012)
- The genetic makeup of the Drosophila piRNA pathway (Handler et al. 2013)
- piRNA-guided slicing specifies transcripts for Zucchini-dependent, phased piRNA biogenesis (Mohn & Handler et al. 2015)
- piRNA-guided slicing of transposon transcripts enforces their transcriptional silencing via specifying the nuclear piRNA repertoire (Senti et al. 2015)
- Genetic and mechanistic diversity of piRNA 3'-end formation (Hayashi & Schnabl et al. 2016)
2. piRNA-guided transcriptional silencing and heterochromatin formation
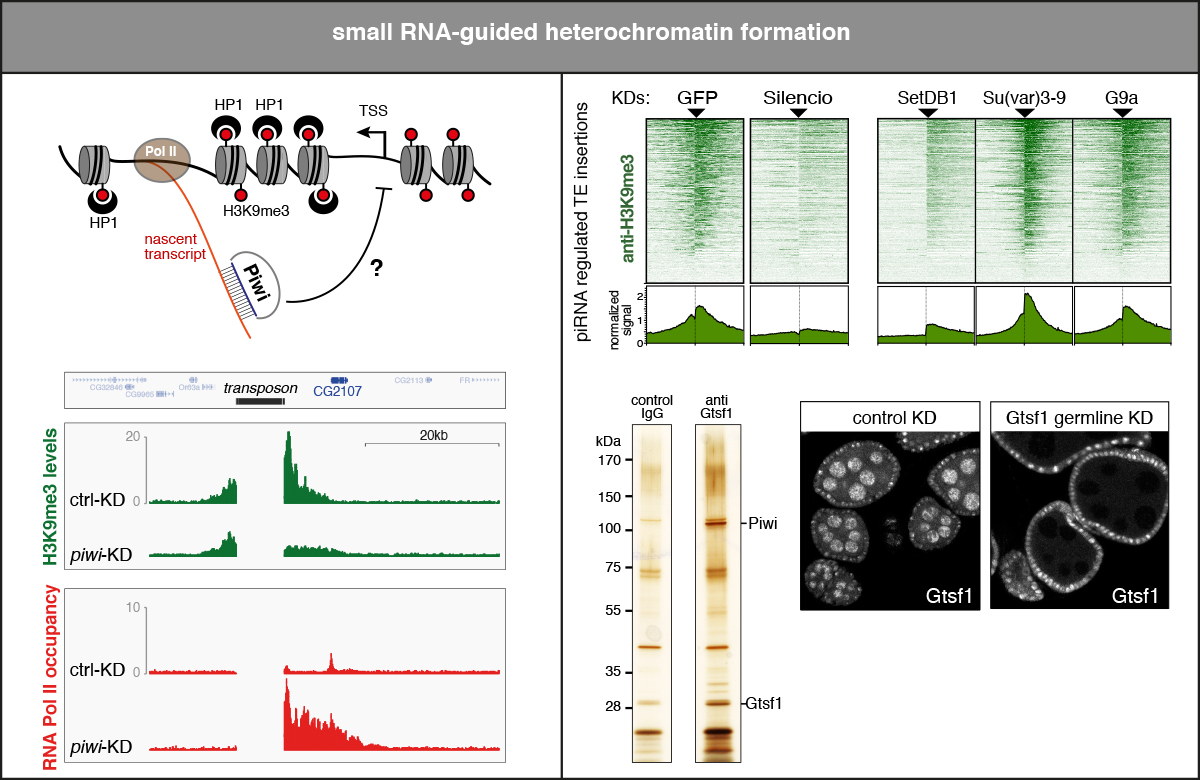
Of the three PIWI-clade proteins in Drosophila, only Piwi is enriched in the nucleus where it targets nascent transposon transcripts and triggers the assembly of a transcriptional silencing complex. This is accompanied by local heterochromatin formation with the key hallmark of H3K9 tri-methylation. Piwi-mediated silencing is a remarkable process as it is independent of target cleavage. How does co-transcriptional recruitment of Piwi to a transposon, which is programmed to be highly transcribed, inhibit gene expression? Which factors are involved, and in which hierarchy do these act?
In many ways, this process resembles transcriptional silencing by KRAB-type Zinc fingers in vertebrates, with the central difference that Piwi is recruited to nascent target RNAs, while KRAB-ZFs operate via DNA targeting. This predicts fundamental differences in the initiation steps of silencing, as one would expect a nascent RNA to be only transiently present at chromatin. A DNA target site instead ‘does not run away’. Equally fascinating is the conundrum of how silencing is maintained, as Piwi targeting both requires and represses nascent RNA. We study piRNA-guided transcriptional silencing and heterochromatin formation in flies and cultured ovarian somatic cells with the aim to understand how Piwi engages the cellular heterochromatin machinery in initiation and maintenance of transcriptional target silencing.
Relevant literature:
- Transcriptional silencing of transposons by Piwi and Maelstrom and its impact on chromatin state and gene expression (Sienski & Donertas et al. 2012)
- Drosophila Gtsf1 is an essential component of the Piwi-mediated transcriptional silencing complex (Donertas & Sienski et al. 2013)
- Silencio/CG9754 connects the Piwi-piRNA complex to the cellular heterochromatin machinery (Sienski & Batki & Senti et al. 2015)
3. The biology of genomic piRNA source loci – hacking the gene expression machinery
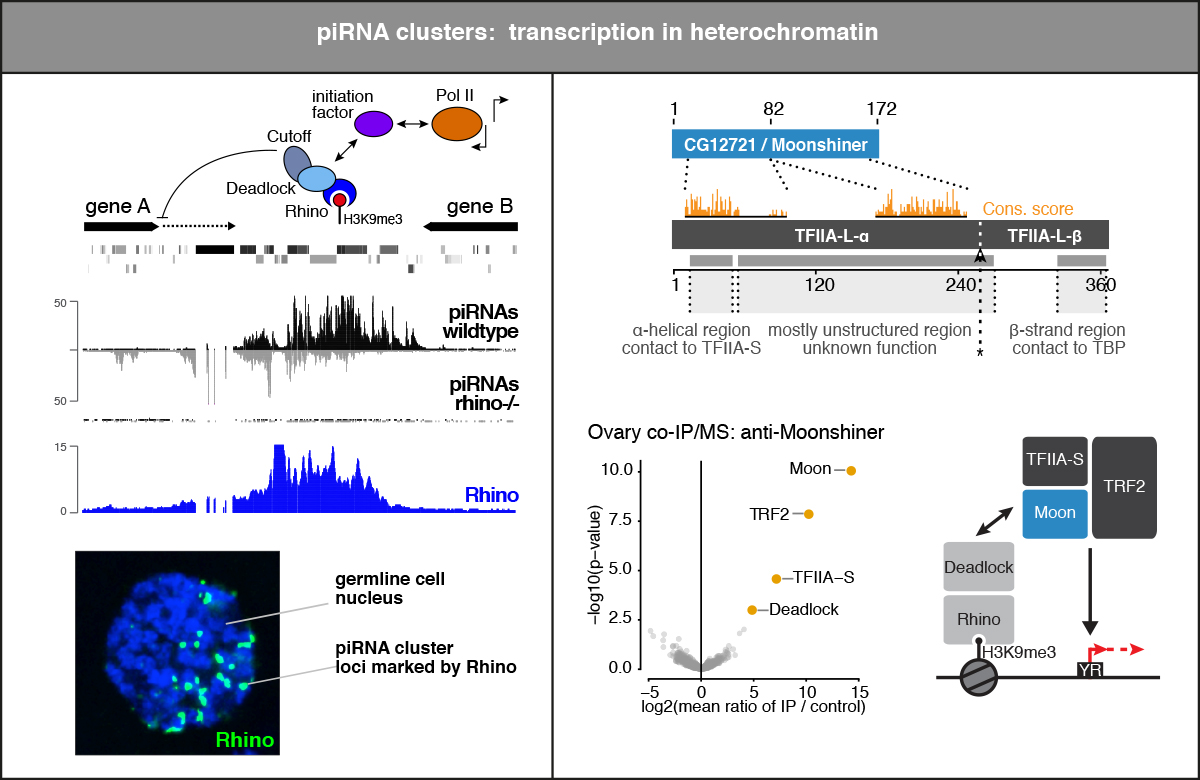
piRNAs are encoded in the genome and recent studies indicate that thousands of genomic loci give rise to piRNAs. Some of these so-called piRNA clusters are clearly special: they encode the majority of all piRNAs and they are enriched in specific subtypes of transposon sequences. For example, the major piRNA cluster in ovarian support cells, flamenco, is almost exclusively composed of sequences of the retroviral gypsy transposons, which can infect the neighboring oocyte. piRNA clusters are therefore the system’s heritable repositories for silencing information. They also adapt to newly invading transposons as transposition into a cluster provides the system with the necessary silencing information.
In the Drosophila germline, most piRNA clusters reside within heterochromatic regions. Perhaps not surprisingly, they are ultimately ideal targets for the nuclear Piwi silencing machinery. But how are they then transcribed in order to fuel the system with piRNA precursors? Recent findings indicate that piRNA clusters are defined by a gonad specific HP1 family protein called Rhino. Rhino recruits a battery of germline specific factors—often sister proteins of ancient gene expression factors—that overwrite the canonical rules of gene expression. These factors cooperate to allow the efficient expression of heterochromatic loci while at the same time canonical transcription from transposon promoters is suppressed. We are interested in the transcription of piRNA clusters as well as in the specification, nuclear export and processing of their transcripts. Our working hypothesis is that by understanding how gene expression can be tricked into functioning at heterochromatic regions, we will also gain fundamental insights into general transcription, RNA processing, and RNA export biology.
Relevant literature:
- The Rhino-Deadlock-Cutoff complex licenses noncanonical transcription of dual-strand piRNA clusters in Drosophila (Mohn et al. 2014)
- A heterochromatin-dependent transcription machinery drives piRNA expression (Andersen et al. 2017)
4. Maternally inherited piRNAs as epigenetic vectors
Drosophila genetics had pointed its finger to key aspects of the piRNA pathway long before RNA interference was discovered. The most famous example is the phenomenon of hybrid dysgenesis, which was described already in the 1970s.
Hybrid dysgenesis occurs when a Drosophila female is mated with a male that harbors a transposable element that is absent in the female’s genome. This cross yields viable, yet sterile offspring. The reciprocal cross yields genetically identical, yet fertile offspring. This must mean that females deposit a key protective agent into the developing oocyte that restricts the activation of the transposon brought in via the male. This maternal contribution is almost certainly piRNAs bound to Piwi and Aubergine. In fact, piRNAs are highly abundant in early embryos, and Piwi as well as Aubergine are enriched in the pole plasm, which specifies the primordial germ cells. Increasing evidence points towards fundamental roles of maternally transmitted piRNAs in the establishment of the embryonic piRNA pathway. First, heterochromatin formation by Piwi specifies target loci not only for silencing, but also licenses them as piRNA source loci by commissioning Rhino recruitment via unknown mechanisms. Second, cytoplasmic cleavage of a piRNA target is required to specify an RNA as a potent piRNA precursor in the germline.
Intriguingly, maternally deposited Piwi is also enriched in every somatic nucleus during and shortly after the cleavage cycles during early embryogenesis. It is very likely that Piwi also contributes to the establishment of heterochromatin domains during this early time point. We aim to identify the first piRNA targets during embryogenesis, to understand when and how piRNA clusters are re-specified during early development, and to dissect the precise roles of maternally inherited Piwi and Aubergine.
Relevant literature:
- An epigenetic role for maternally inherited piRNAs in transposon silencing (Brennecke & Malone et al. 2008)
5. Venturing into mammals
The piRNA pathway described above, and homologous pathways in other organisms (i.e. plants), ensure a tight control of the transposon landscape of the genome. However, little is known about how newly colonizing transposons, with no homology to the small RNA repertoire of the host, are detected and set for silencing. Are specific features of transposon DNA/RNA recognized? Does their unique ability to integrate new copies of themselves into the genome trigger silencing? In recent years, embryonic stem cells (ESC) have become a key model for mammalian chromatin biology and epigenetics, uncovering a complex interplay between transposons and key developmental processes during early embryogenesis. Although many aspects about how repressive chromatin marks are maintained at transposons and other repetitive sequences have been characterized recently (e.g. HUSH/Morc2 biology), the initial trigger(s) for true de novo silencing remain obscure. The combined power of ES cells haploid genetics and CRISPR/Cas driven genetic screens opens an unprecedented opportunity to understand the mechanisms that lead to the initial recognition and transcriptional silencing of foreign DNA elements.