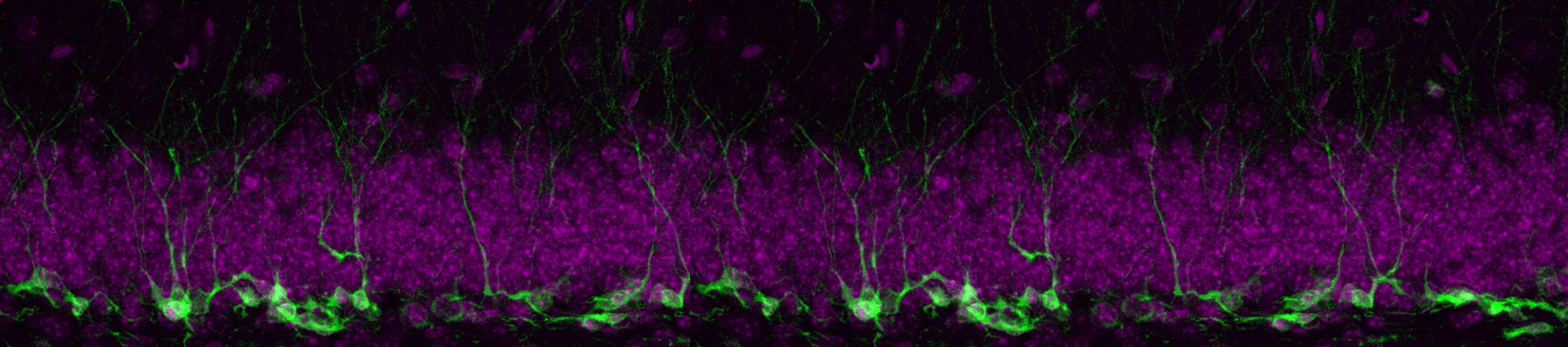
IDENTIFYING SIGNALS THAT CONTROL NSC QUIESCENCE
Adult neurogenesis can be modulated by a great variety of local signals and systemic stimuli, such as diet or stress. However, the molecular mechanisms regulating adult NSCs are largely unknown. We use novel in vivo and in vitro tools to study the response of NSCs to different stimuli. Our current focus is on signaling cues directly reaching adult NSCs, including systemic signals and those coming from the extracellular matrix. We give particular importance to testing the effects of these stimuli on the maintenance of the stem cell pool, therefore allowing us to determine their long-term consequences on adult neurogenesis and behaviour.
Adult NSCs are exposed to multiple, often opposing, signals at the same time. We are interested in the internal signal integration mechanisms present in adult NSCs that allow them to mount an appropriate response to their complex signaling environment. For this, we are very lucky to have recently teamed up with the SMICH community (further details available soon here).
One of the most important drivers of the activation of adult NSCs is the transcription factor Ascl1. We know Ascl1 protein levels are regulated by the E3-ubiquitin ligase Huwe1 and that this regulation is behind the changes in adult NSC behaviour observed during ageing. One of our most active projects aims at identifying new regulators of the quiescence state by examining how the Huwe1-Ascl1 axis is controlled. This project has been boosted by a SFB collaborative grant, with which we are also investigating what is the role of targeted protein degradation in the quiescence to activation transition. Please check the Targeted Degradation website for more information.
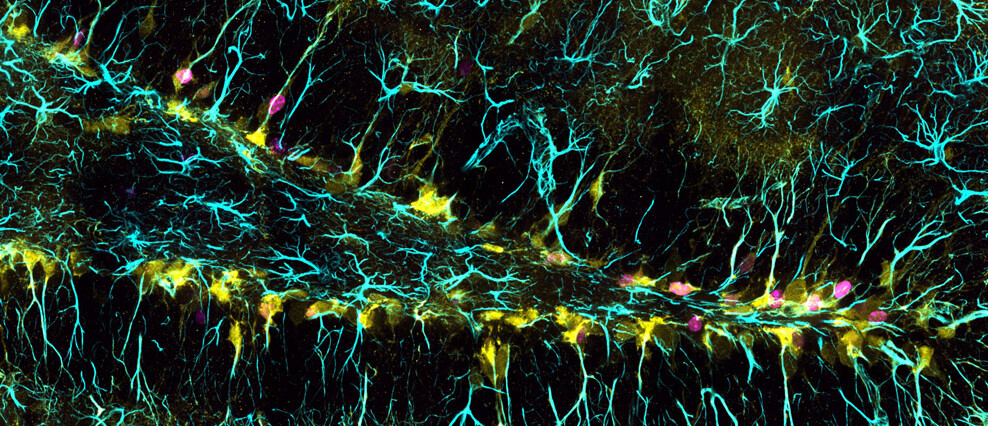
EFFECTS OF SYSTEMIC STIMULI ON ADULT NEUROGENESIS
We use lineage tracing and label retention experiments to characterize the response of adult neural stem cells to “western/cafeteria" diets or dietary restrictions. Western or cafeteria diets with a high content of fat and sugar are key players behind the emergence of a type 2 diabetes epidemic worldwide and they have been shown to decrease adult neurogenesis. On the other hand, dietary restrictions such as calorie restriction or intermittent fasting are seen as “rejuvenating" diets with the potential to prevent metabolic diseases and delay aging. However, the long-term consequences of these diets on adult NSCs and therefore the lasting repercussion on adult neurogenesis have not been studied yet. Our goal is to characterize the relationship between diet and the regulation of adult neural stem cells to broaden our understanding of the impact of diet on adult neurogenesis, memory and behaviour.
We are also interested in finding the molecular effectors underpinning the modulation of adult neurogenesis by diet. Our current focus is to decipher how insulin and IGF, which are systemically changed upon dietary interventions, affect adult NSC behaviour.
Thanks to a SFB collaborative grant, we are investigating how age affects the response of adult NSCs to systemic cues and whether they are regulated by circadian rhythms. More details will be available soon on the Neuromodulation Consortium website.
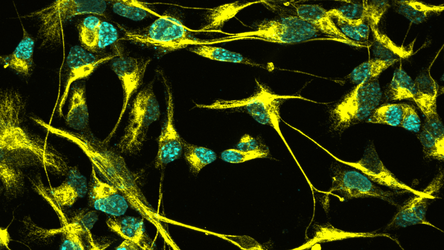
GENERATING A MODEL OF HUMAN NSC QUIESCENCE
We still know very little about adult NSCs in humans, and there is currently a heated debate as to whether hippocampal neurogenesis is present only in young individuals or persists throughout adulthood. We are currently generating a quiescence model using hiPSCs in order to start exploring the regulation of quiescence in human neural stem cells. In collaboration with the Edenhofer lab in Innsbruck, we will compare the behavior of reprogrammed NSCs (from iPSCs) to that of directly converted NSCs (from adult fibroblasts), which will allow us to elucidate the effects of ageing on NSC quiescence. More details will be available soon on the Neuromodulation Consortium website.
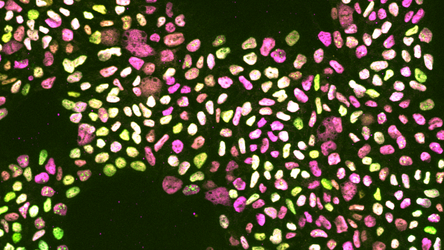
UNTANGLING QUIESCENCE FROM STEMNESS
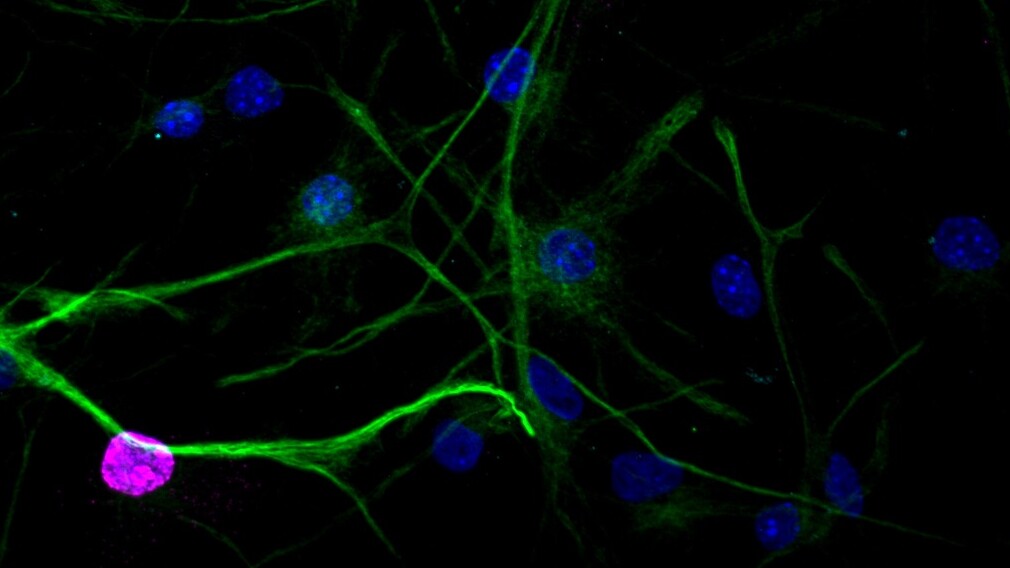
GFAP (green), Ki67 (magenta) and DAPI (blue)
While in the developing brain, proliferation is associated with the retention of stem cell characteristics, this is quite the opposite in the adult brain. Adult NSCs actually lose their self-renewing capacity upon successive divisions. We hypothesize that quiescence-related signals not only keep NSCs out of the cell cycle, but also allow them to retain their stemness. We are exploring how quiescence-inducing signals affect the self-renewing capacity of NSCs. It has been reported that NSCs gradually switch mode of division, from symmetric self-renewing to asymmetric and eventually to symmetric differentiating. To determine whether quiescence can also influence the mode of division of NSCs, we are currently devising strategies to quantify asymmetric vs symmetric divisions in NSCs using live imaging techniques.